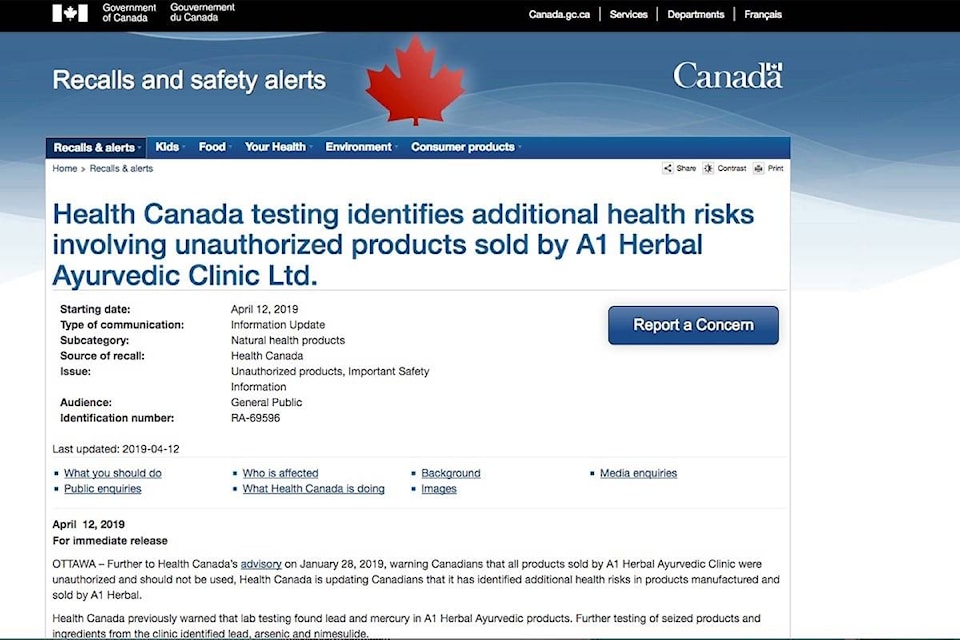
A Surrey herbal clinic is once again the subject of a Health Canada warning.
If you have any products from A1 Herbal Ayurvedic Clinic Ltd. (31-8430 128th St. in Newton), including through its website, Health Canada advises you to “stop using them immediately.”
The Now-Leader tried to contact the clinic on Wednesday, but its phone number was out of service.
This latest Health Canada advisory is dated April 12.
“Consult your health care professional if you have used any A1 Herbal Ayurvedic products and have health concerns,” it states. “Properly dispose of the products according to municipal waste guidelines. Report any health product-related adverse reactions or complaints to Health Canada.”
The first health warning was issued on Jan. 28 after the BC Centre for Disease Control advised the public to discard products purchased from the clinic after they were found to contain lead and mercury.
READ ALSO: Health warning issued on Surrey herbal product
Fraser Health issued a closure order pursuant to the Public Health Act, after an inspection, and seized products, ingredients and equipment.
The latest advisory indicates that further testing has also found arsenic and nimesulide.
“Arsenic, lead and mercury are heavy metals that pose serious health risks,” the advisory notes. “Ingestion of excessive amounts of arsenic, lead or mercury may accumulate in vital organs of the body. Children are most susceptible to the toxic effects of heavy metals. Nimesulide is a drug that is not authorized for sale in Canada and has been withdrawn from the market in several countries because it is associated with liver damage.”
Ayurvedic medicinal products are used in traditional Indian healing practice, and are often imported from India.
Of affected products, the latest advisory indicates, Fat Go Super Slim, Ashoka 50%, Punarua 50%, C-24 500 mg, KAM Extract, and Triffla 50% are contaminated with lead.
ARS K RAS, Chirita 50% and Amba Haldi 50% contain arsenic, lead and nimesulide, and Daru HLDI 33%, Punarua 33%, Amla 33%, DESI Extract, Giloy, Tulsi and K-Guggal Extract contain nimesulide.
tom.zytaruk@surreynowleader.com
Like us on Facebook Follow us on Instagram and follow Tom on Twitter